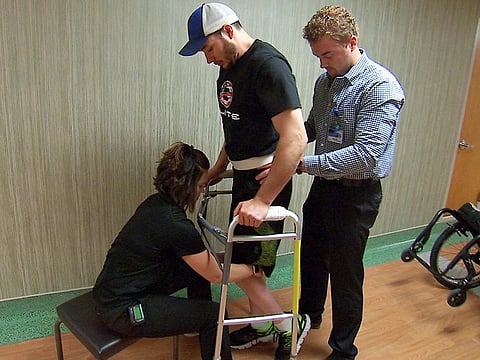
MONDAY, Sept. 24, 2018 (HealthDay News) -- A paraplegic man has regained the ability to move his legs and walk with assistance, thanks to an implanted electrode stimulating his spinal cord, Mayo Clinic researchers say.
Surgeons implanted the electrode below the level of 29-year-old Jered Chinnock's spinal cord injury. A 2013 snowmobile crash left Chinnock with complete loss of motor control and sensation below the middle of his back.
But after the new therapy, he "was able to regain voluntary control of the movement in his legs," said co-principal investigator Dr. Kendall Lee, a neurosurgeon and director of Mayo Clinic's Neural Engineering Laboratories in Rochester, Minn. He explained that Chinnock's "own mind or thoughts were able to drive the movement in the legs."
Similar results were also reported Monday for patients who got the same type of treatment in a study conducted at the University of Louisville.
Describing Chinnock's case, researchers said he can now can walk about the length of a football field, around 111 yards.
"We were able to get him to stand independently and be able to take his own steps," Lee said. "The amount of steps he was able to take was pretty significant."
New insights into the spinal cord
Researchers aren't sure why this electrical stimulation allows the brain to once again take control of the legs, Lee said.
He noted that the electrode is placed "way below the level of the injury," stimulating nervous tissue that still is connected to the leg muscles.
It's possible that despite the injury, there remain some residual intact nerve fibers capable of transmitting brain signals to the legs, said Kristin Zhao, co-principal investigator and director of Mayo Clinic's Assistive and Restorative Technology Laboratory.
If that's the case, the brain likely is sending signals to re-stimulated nerves farther down the spinal cord that are specifically tied to walking, said Dr. Brian Kopell, a neurosurgeon and director of the Center of Neuromodulation at Mount Sinai Health System in New York City.
"We are beginning to understand there are specific hard-wired circuits related to walking in the spinal cord itself," said Kopell, who wasn't involved with the study. "The brain works in conjunction with these locomotive sectors in the spinal cord to create the behavior we know as walking."
How it works
This study began in 2016, with Chinnock receiving his electrode implant after 22 weeks of physical therapy.
It sits in the epidural space that covers the spinal cord, Lee said. It is connected to a pulse generator implanted just below the skin of his abdomen.
Researchers can wirelessly program the pulse generator to provide specific electrical stimulation to the spinal cord, Lee said.
After recovering from surgery, Chinnock had 43 weeks of intense physical therapy involving 113 visits to the Mayo Clinic, Zhao said.
He eventually regained the ability to walk over ground using a front-wheeled walker and on a treadmill with his arms on support bars to help with balance.
By the end of the study period, Chinnock learned to use his entire body to transfer weight, maintain balance and propel himself forward, researchers said.
But his legs move only when the pulse generator is activated, Lee said.
"The stimulation absolutely has to be turned on," Lee said. "We found you have to deliver a very specific type of stimulation. A random stimulation does not work."
Chinnock also still cannot feel anything below the site of his spinal cord injury, Lee added.
He cannot yet walk independently outside the lab, but does perform regular leg exercises at home while standing or sitting, Zhao said.
Full independence the goal
Chinnock said the implant also has helped him in one of his favorite pastimes, bowhunting.
"My sitting balance and stuff has gotten a lot better. Like, I can shoot my bow a lot better because I'm able to hold -- have more trunk support and stuff," he said in a video released by Mayo.
Chinnock said his goal is to become "completely independent -- be where I needed a walker, but I didn't need anybody else to help me. I mean, that's kind of a goal, but the main goal is to not need anything."
The electrical stimulator he has is one designed for nerve pain. The research team received permission from the U.S. Food and Drug Administration to use it in this new way.
Researchers now plan to step back and re-engineer the device to specifically target paralysis, Lee said.
They also plan more studies to figure out just what is happening in the brain and spinal cord that allows a patient to regain control of his legs, Zhao said.
The latest report on this study appears in the journal Nature Medicine.
In a similar separate study published this week in the New England Journal of Medicine, University of Louisville researchers reported that two of four paralyzed patients were able to walk again after receiving an implanted stimulation device and intense physical therapy.
Susan Harkema, a professor of neurological surgery who was part of that research, described the outcome as "phenomenal" in an interview with CNN.
"This new knowledge is giving us the tools to develop new strategies and tools for recovery in people with chronic spinal injuries," she said.
Harkema and her colleagues implanted epidural stimulators in 14 paralyzed patients over the years. Thanks to the devices, all 14 were able to move and had better bowel and bladder function, she said.
"This should change our thinking about people with paralysis," Harkema said.
More information
Columbia University has more about neuromodulation.